Do you suffer from pain, tingling, burning, or numbness from your chemotherapy treatment?
You may qualify for a clinical trial of an investigational treatment for chemotherapy-induced pain.
What is CINP?
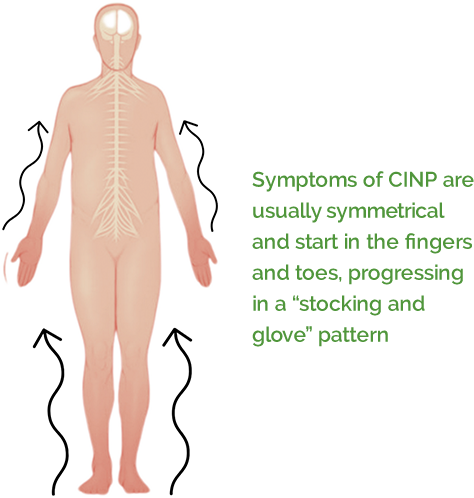
Chemotherapy and other cancer treatments can sometimes damage nerves in the extremities – such as the fingers, hands, arms, feet, and legs. This condition is known as chemotherapy-induced nerve pain, or CINP.
Some types of chemotherapy are more likely to cause nerve damage than others and experts believe CINP is experienced by at least 30% – 60% of people who receive these types of chemotherapy. This can occur in patients undergoing treatments for many types of cancer including breast, colon, lung, prostate, etc.
If you have previously undergone chemotherapy and now struggle symptoms including, pain, tingling, electric shocks, burning or numbness in your hands, arms, feet, or legs, you may qualify to participate in a new clinical trial. The trial is for Halneuron®, an investigational medication being evaluated for the treatment of neuropathic (nerve) pain due to previous taxane or platinum chemotherapies. If you qualify and choose to participate you will receive all study-related care at no cost.
About the Study
Doctors at select centers in the US are accepting new participants for a clinical trial of Halneuron®, an investigational medication being evaluated for the treatment of neuropathic (nerve) pain due to previous taxane or platinum chemotherapies. If you have undergone taxane or platinum chemotherapy and now struggle with nerve pain, you may be eligible to participate.
Halneuron is delivered as an injection just under the skin, and is performed at your doctor’s office. In this study, Halneuron will be compared to a placebo, a treatment that does not contain any active drug. Study participants will receive once-a-day injections of either Halneuron or placebo 8 times over a 2-week period. You will then return to your study doctor’s office 2 weeks later to complete certain tests and questionnaires to evaluate your progress. Your total participation in the study will last from 5 – 9 weeks.
If you qualify and choose to join the study, you will receive all study-related care and study medication at no cost, and will also be compensated for your time and travel to attend any required visits.

See If You Qualify
To see if you might qualify for the study, and to get connected with your local study center, please complete the questionnaire below:
